Nagoya University researchers used iron and blue LEDs to synthesize natural molecules, cutting the need for expensive chiral components by two-thirds.
Photocatalysts facilitate chemical reactions by absorbing light. Metal-based photocatalysts are widely used in organic synthesis due to their durability and the ability to tune their function by modifying the ligands attached to the central metal atom.
Most metals used in photocatalysts, such as ruthenium and iridium, are rare and expensive. Researchers at Nagoya University, Japan, previously developed an iron-based alternative, but it required large amounts of costly chiral ligands, which act as spatial templates to determine the three-dimensional structure of chemical products.
In a recent study published in the Journal of the American Chemical Society, the researchers developed an iron catalyst that reduces the use of chiral ligands by two-thirds and enables photocatalytic reactions under energy-efficient blue LED light.
Using this new catalyst, they completed the asymmetric total synthesis of (+)-heitziamide A, a natural compound from medicinal plants that suppresses respiratory bursts.
Professor Kazuaki Ishihara, Assistant Professor Shuhei Ohmura, and graduate student Hayato Akao at Nagoya University’s Graduate School of Engineering developed this technology.
Redefining the design of iron catalysts
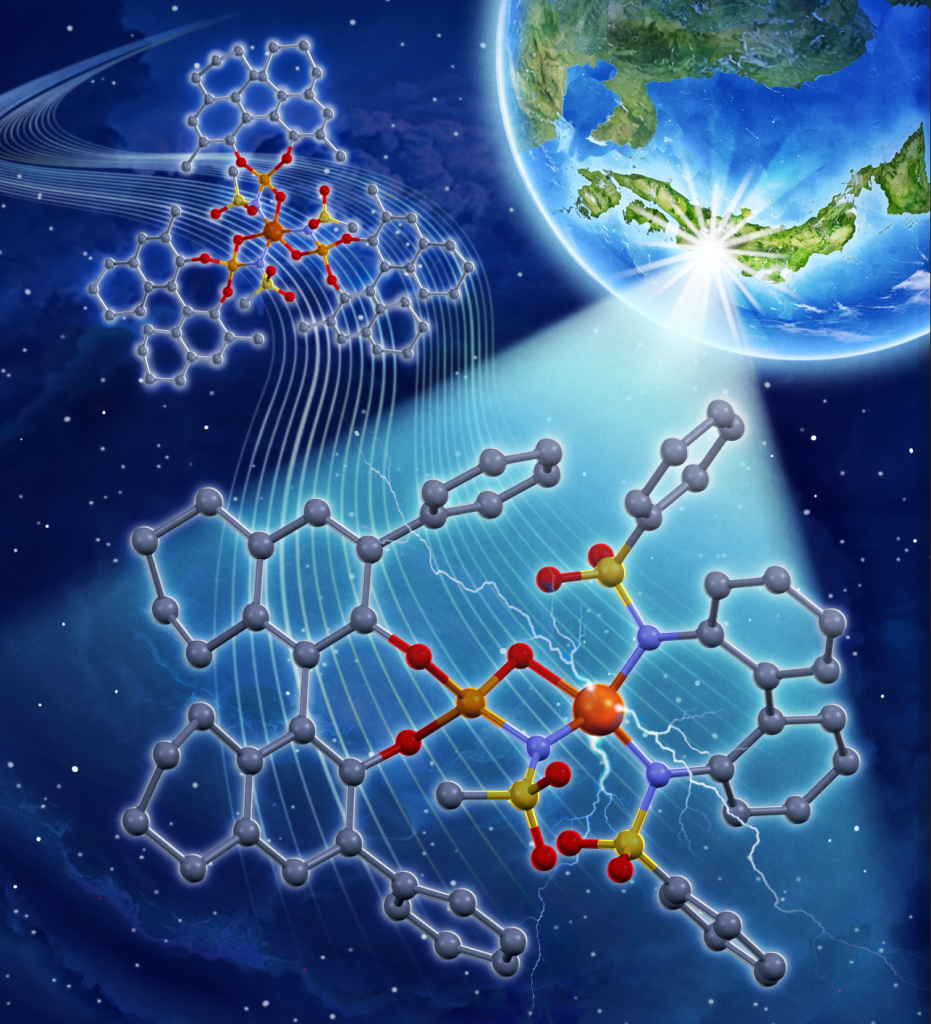
In their 2023 study, the researchers developed an iron photocatalyst that used three chiral ligands per iron atom, but only one-third of these ligands contributed to enantioselectivity, making the process inefficient.
Meanwhile, the newly developed iron photocatalyst combines cost-effective achiral bidentate ligands with chiral ligands to target a specific iron(III) salt structure. The chiral ligand controls the three-dimensional configuration, while the achiral bidentate ligand tunes the catalytic activity.
Using this catalyst, researchers achieved a precise radical cation (4 + 2) cyclization, joining two molecules to form a hexagonal ring. This method enables the synthesis of 1,2,3,5-substituted adducts, structures common in natural products such as heitziamide A.
“The new catalyst design represents the definitive form of chiral iron(III) photoredox catalysts,” stated Ohmura, one of the study’s corresponding authors. “We believe this achievement marks a significant milestone in advancing iron-based photocatalysis.”
Advancing artificial synthesis of (+)-heitziamide A
While artificial synthesis of heitziamide A has been previously reported, the total asymmetric synthesis of its natural enantiomer has not yet been achieved.
Using selective six-membered-ring formation with an iron photocatalyst activated by blue light, the researchers achieved the first total asymmetric synthesis of (+)-heitziamide A. This indicates that using the mirror-image catalyst would also allow the synthesis of (-)-heitziamide A, thereby enabling the selective production of both enantiomers.
Significance and future perspectives
The newly developed iron photocatalyst enables the precise synthesis of complex molecules, including pharmaceutical precursors, using abundant iron and blue LEDs instead of rare metals.
“Achieving the first-ever asymmetric total synthesis of (+)-heitziamide A using this catalytic reaction is a remarkable accomplishment,” stated Ishihara, the study’s other corresponding author. “Several additional bioactive substances can be accessed through total synthesis, with enantioselective radical cation (4 + 2) cycloaddition serving as a key step. We intend to publish follow-up papers on the asymmetric total synthesis of these compounds in the near future.”
Paper information:
Hayato Akao, Shuhei Ohmura, and Kazuaki Ishihara (2026). A Rational Design of Chiral Iron(III) Complexes for Photocatalytic Asymmetric Radical Cation (4 + 2) Cycloadditions and the Total Synthesis of (+)-Heitziamide A, Journal of the American Chemical Society. DOI: 10.1021/jacs.5c20243
Funding information:
This work was supported by JSPS KAKENHI grants 24K17677 and 23H05467.
Expert contact:
Kazuaki Ishihara
Graduate School of Engineering, Nagoya University
ishihara.kazuaki.s7@f.mail.nagoya-u.ac.jp
Media contact:
Naomi Inoue
International Communications Office, Nagoya University
icomm_research@t.mail.nagoya-u.ac.jp
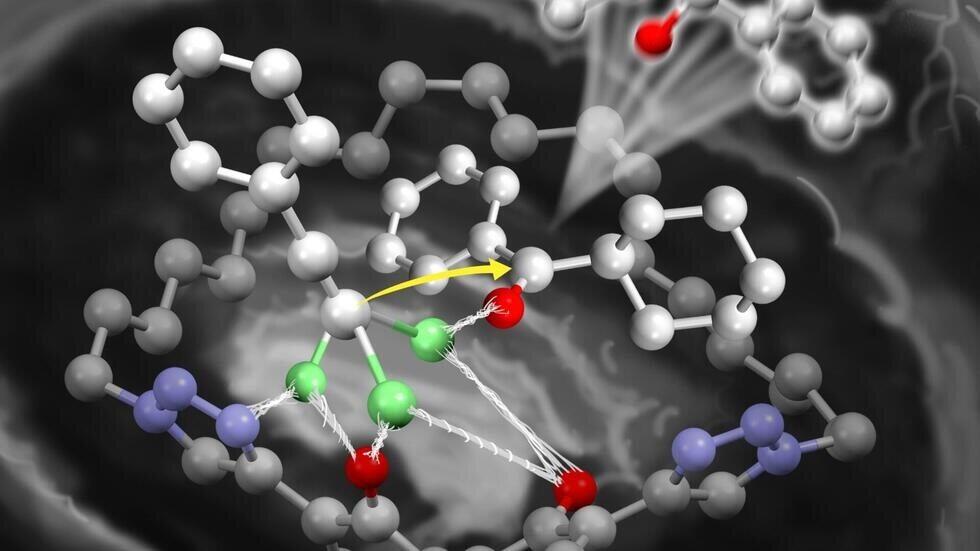
Top image:
The newly designed iron photocatalyst (front) and the previous catalyst (back)
(Credit: Yuzuru Endo)