Fumagillin has been investigated as a drug lead for more than 70 years, but its more potent relative ovalicin was never developed. Now, scientists have identified metabolic instability as the key barrier and have overcome it through chem-bio hybrid synthesis, yielding drug candidates for amebiasis, a parasitic infection affecting 50 million people annually.
Amebiasis is a parasitic disease caused by the microscopic protozoan Entamoeba histolytica. Infection occurs through the ingestion of cysts from contaminated water or food. Worldwide, approximately 50 million symptomatic cases are estimated annually, mainly in tropical and subtropical regions.
Fumagillin, a fungal natural product, has been studied for decades as a potential antiparasitic drug, but its more potent relative ovalicin was never developed. Now, a study published in the Journal of the American Chemical Society reveals why: although ovalicin is highly active against amebiasis, liver enzymes rapidly break it down in the body. Researchers used a chem-bio hybrid approach to turn that insight into metabolically stable drug candidates that worked in animal models of amebiasis, including liver infection with abscess formation.
The research team, led by scientists from the Graduate School of Bioagricultural Sciences at Nagoya University, identified the liver cytochrome P450 enzymes responsible for ovalicin breakdown, with CYP 2B1 and CYP 2C6 emerging as the main drivers. Blocking these enzymes with a chemical inhibitor significantly prolonged ovalicin survival, providing strong evidence that rapid liver metabolism limits its effectiveness.
Successfully curing infections in animals
“We engineered fungi to build modified ovalicin molecules with a special attachment point that we could customize. We then clipped different molecular groups onto that point to create versions that the liver could not destroy,” explained senior author and associate professor Yuta Tsunematsu.
Using genetically engineered filamentous fungi, the team produced gram-scale quantities of a non-natural ovalicin molecule. They then created about 30 derivatives and tested each one to find versions that killed parasites, survived liver breakdown, and were not toxic.
The protozoan parasite depends on an enzyme called MetAP2 for its proteins to work properly and survive. Blocking MetAP2 kills it but does not harm humans because we have a backup enzyme that can perform the same function.
Two of the new compounds, YOK24 and NS-181, blocked the parasite’s MetAP2 enzyme and eliminated the parasitic infection in hamsters, causing liver abscesses to disappear entirely.
These results are an important step toward testing these drug candidates in humans. Importantly, the compounds were effective after both injection and oral administration. Oral treatment would be especially valuable in low-resource settings, where amebiasis is most prevalent.
A new drug development method
Current amebiasis treatments, such as metronidazole, can cause side effects and face growing concerns about drug resistance.
This study introduces Chem-Bio Hybrid Synthesis, a new method that combines genetic engineering of microbes and chemistry to transform natural compounds that worked in lab tests but failed in patients due to rapid breakdown or toxicity. The approach could address these challenges for amebiasis and be applied to develop treatments for other parasitic diseases, cancer, and obesity.
Paper information:
Yuki Okura, Yumiko Saito-Nakano, Andrii Balia, Nurul Syahmin Binti Suhaimi, Chika Ando, Namiko Ogata, Tomona Ikeda, Takumi Sato, Keiko Kano, Emi Mishiro-Sato, Masaki Kita, Noriyuki Miyoshi, Kenji Watanabe, Kouichi Yoshinari, Norio Shibata, Mihoko Mori, Seiki Kobayashi, Yuji Sumii, Ryota Shizu, Tomoyoshi Nozaki, Yuta Tsunematsu (2026). Chem–Bio Hybrid Synthesis Enables Reengineering of Natural Product-Based Methionine Aminopeptidase 2 Inhibitors for Treating Amebiasis, Journal of the American Chemical Society, 148(7), 7189–7201. DOI: https://doi.org/10.1021/jacs.5c18554.
Funding information:
This work was financially supported by the Japan Agency for Medical Research and Development (AMED) (Grant numbers: JP22wm0325020, JP23wm0325070, JP25jm0110022) and the Japan Society for the Promotion of Science (JSPS) (Grant number 24K02190).
Expert contact:
Yuta Tsunematsu
Graduate School of Bioagricultural Sciences
Nagoya University
E-mail: tsunematsu.yuta.p4@f.mail.nagoya-u.ac.jp
Media contact:
Merle Naidoo
International Communications Office
Nagoya University
Email: icomm_research@t.mail.nagoya-u.ac.jp
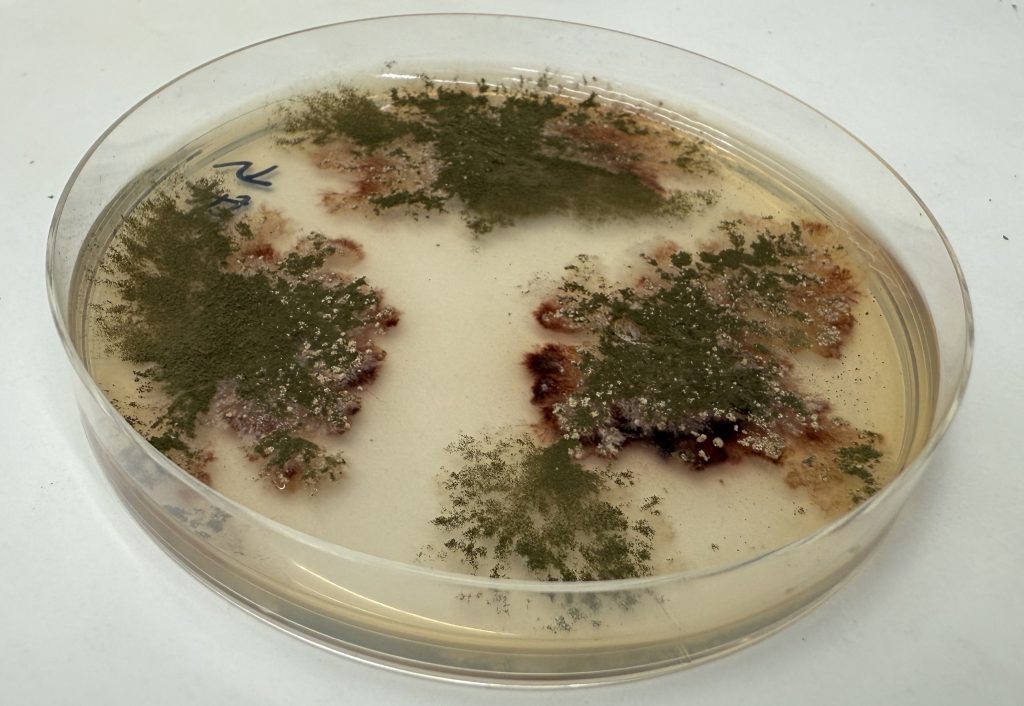
Top image:
Scientists genetically engineered the fungus Aspergillus nidulans to produce a modified ovalicin molecule. Although this molecule itself was still susceptible to liver breakdown, it provided a chemical handle that enabled the synthesis of metabolically stable drug candidates for amebiasis. Credit: Yuta Tsunematu, Nagoya University