Nagoya University-led study shows for the first time germ cells have an inherent property to feminize the body in teleost fish, medaka.
press released on March 29, 2018
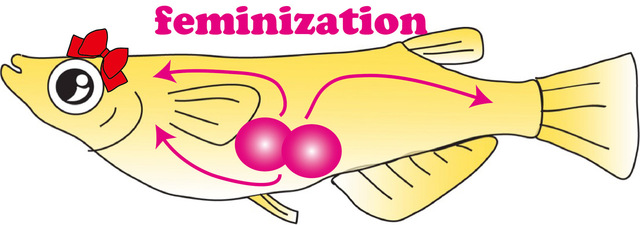
Fig. 1. Identification of cells that determine the female gender in teleost fish (medaka).
The pink circles represent germ cells (the common origin of sperm and eggs). Germ cells, irrespective of the sexual fate (to become sperm or eggs), have the potential to feminize the body in medaka. The pink arrows indicate the feminizing effect from germ cell. (© Toshiya Nishimura)
Nagoya, Japan - The gender of living organisms is determined either by genetic factors and/or by environmental factors. Interestingly, however, a team of researchers led by Nagoya University previously found that the occurrence of more germ cells - biological cells capable of uniting with one from the opposite sex to form a new individual - in female medaka (teleost fish) gonads is essential for female differentiation of gonads. When germ cells are removed in medaka, XX (female) fish show female-to-male sex reversal, while XY (male) fish with excessive germ cells, which is usually associated with egg production, exhibit male-to-female sex reversal.
"This finding implies that, in addition to the most well- known role of germ cells developing into eggs or sperm and produce the next generation, germ cells have a unique and surprising potential to change their surrounding environment," says Toshiya Nishimura, first author of the current study. "However, the molecular basis of, and the stage of gametogenesis critical for, feminization remain unknown."
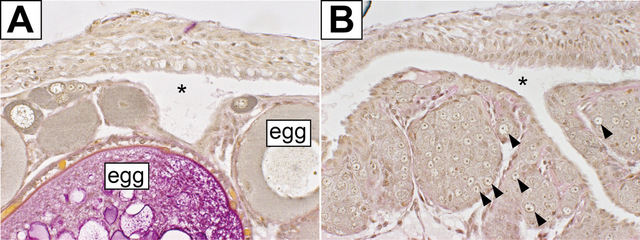
Fig.2. The feminizing effect is inherent to germ cells which are essential for the formation of ovaries.
(A) Wild-type ovary which is occupied with many eggs. The asterisk indicates the ovarian cavity, where ovulation of mature eggs occurs. The ovarian cavity represents the structure of ovaries. (B) Mutant ovary with ovarian cavity (asterisk) without eggs. In this ovary, only sexually indifferent germ cells are present (arrowheads). Due to the feminizing effect of these germ cells, medaka can form ovaries and become female. (© Toshiya Nishimura, modified from Nishimura et al., 2018)
This question set the researchers to delve deeper into the topic. In their latest study, they generated three different medaka mutants to demonstrate that the feminizing effect of germ cells is not a result of the progression of gametogenesis or a sexual fate decision of germ cells. They found the different stages of germ cells in XX mutants have an ability to feminize the gonads, resulting in the formation of gonads with ovarian structures. In addition to normal ovarian development, the increased number of gonocytes (fetal and neonatal germ cells) is sufficient for male-to-female sex reversal in XY medaka.
What these results mean is that the mechanism underlying the feminizing effect of germ cells is activated before the sexual fate decision of germ cells and meiosis, probably by the time of gonocyte formation in medaka. The study was published in PLOS Genetics.
"Our analyses using medaka collectively also suggest that germ cells acquire the feminizing effect before committing to gametogenesis, while male germ cells undergo a quiescent state before the initiation of spermatogenesis," corresponding author Minoru Tanaka says. "Why do male germ cells need to be quiescent? One possible answer for the biological meaning of the quiescent state of male germ cells may be to prevent the gonad from being feminized until masculinization of somatic cells is established."
*****
The article, "Germ cells in the teleost fish medaka have an inherent feminizing effect", was published in PLOS Genetics at DOI: 10.1371/journal.pgen.1007259.
This work was done in collaboration with Dr. Noriyoshi Sakai and Dr. Toshihiro Kawasaki at National Institute of Genetics and Dr. Kellee R. Siegfried at University of Massachusetts Boston.
*****
Authors: Toshiya Nishimura1, Kazuki Yamada1, Chika Fujimori1, Mariko Kikuchi1, Toshihiro Kawasaki2,3, Kellee R. Siegfried4, Noriyoshi Sakai2,3, Minoru Tanaka1
1 Division of Biological Science, Nagoya University
2 Genetic Strains Research Center, National Institute of Genetics
3 Department of Genetics, SOKENDAI (The Graduate University for Advanced Studies)
4 Biology Department, University of Massachusetts Boston
*****
Related Links:
- Lab. of Prof. Tanaka and Assistant Prof. Nishimura, Division of Biological Science, Graduate School of Science, Nagoya University
*****
Media Coverage:
- EurekAlert!
- AlphaGalileo
- Asia Research News
- Phys.org
- Bioengineer.org
- Scienmag
- Bright Surf
- Science Daily
- SSRIdrugs
- Broken Planet
- Medicine news line
*****
Funding: This work was supported by a Grant-in-Aid for Research Activity Start-up (15H06838:TN), a Grant-in-Aid for Young Scientists (B) (16K18557:TN), a Grant-in-Aid for Scientific Research on Innovative Areas (17H06430: MT), a Grant-in-Aid for Scientific Research (A) (16H02514: MT).